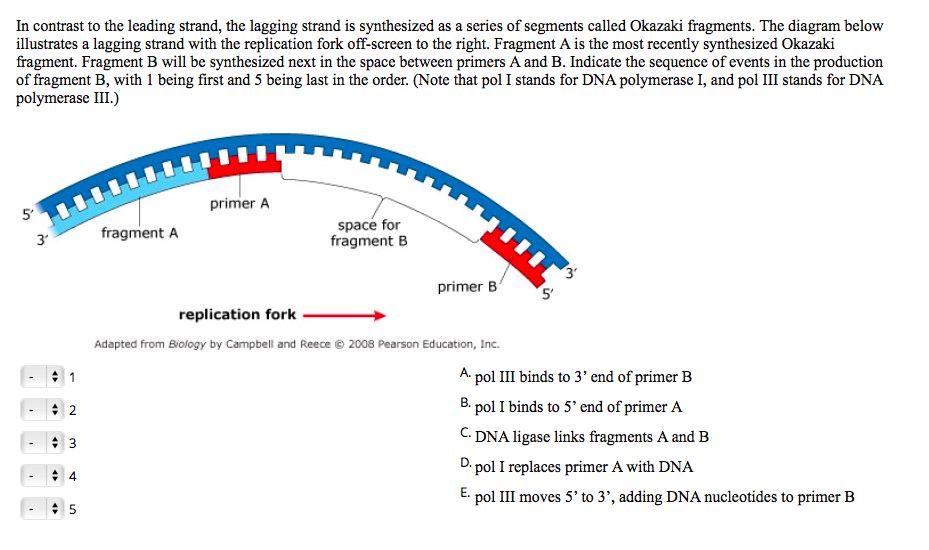
Their sequencing data revealed 28 somatic mutations that distinguished the two cells, including changes in mitochondrial DNA. The researchers used the new approach to sequence DNA from two different T-cells obtained from the same person. 
Compared to short-read sequencing, the new method captured four times as many structural variants-large rearrangements of DNA-including those located in the genome’s inaccessible “dark regions.” The researchers performed the droplet-based amplification on individual human T-lymphocytes, then generated long reads using PacBio HiFi technology. “There is a more even amplification, so you get better representation of the genome,” Adam Ameur, a bioinformatician at Uppsala University in Sweden, tells The Scientist. It works by trapping DNA fragments in droplets that contain a limited supply of reagents, preventing over-amplification of certain regions. To minimize amplification bias, the team used a technique called droplet-based multiple displacement amplification. See “ Large Scientific Collaboration Aim to Complete Human Genome” Though not yet peer-reviewed, the results were reported in a preprint uploaded to bioRxiv on January 23. Now, Hård and colleagues have obtained long reads from individual cells using an improved DNA amplification method. “So substantial amplification is required before you can sequence it” using long-read methods, she says.Īnd that’s where things get tricky, says Hård, as the primary methods used to amplify DNA are prone to “amplification bias”: the tendency for certain sequences to be ramped up at the expense of others. Several micrograms of genetic material are needed, but “a single cell contains just six picograms,” says Joanna Hård, a computational biologist at ETH Zurich in Switzerland. However, long-read sequencing requires a ton of DNA. Long-read sequencing has allowed scientists to sequence troublesome “dark regions” of the genome that are inaccessible to short-read technologies, either due to an abundance of guanines and cytosines, or duplicated regions not easily mapped to a chromosome. Over the past decade, reads-the product of DNA sequencing-have been getting longer. Alexander Hoischen, Radboud University Medical Center The feat may allow for a more detailed look at mutations underlying all sorts of diseases, experts tell The Scientist. “This was unthinkable just two or three years ago,” says Hoischen. By combining “two very innovative approaches”-a cutting-edge DNA amplification technique with the latest advances in DNA sequencing-a team of scientists have applied long-read technology to single cells, says Alexander Hoischen, a researcher of genomic technologies at Radboud University Medical Center in The Netherlands who was not involved in the research. Single-cell, long-read sequencing has remained frustratingly out of reach. Long-read technologies could overcome this pitfall however, their methods require much more DNA than can be extracted from a single cell. But current methods require reading the genome in short sections and therefore often fail to capture complex repetitive regions, which scientists are increasingly linking to health and disease. More recently, scientists have gained the ability to perform single-cell sequencing, which can reveal rare variations between cells and the evolution of cell lineages. Traditional sequencing is often likened to making a smoothie: researchers blend a bunch of cells, obtain an average sequence, and draw conclusions on the ingredients that comprise the slush. 
ABOVE: MODIFIED FROM © ISTOCK.COM, KOBEE & ENOT-POLOSKUN
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
